Search results
Search for "molecular switches" in Full Text gives 65 result(s) in Beilstein Journal of Organic Chemistry.
Synthesis of photo- and ionochromic N-acylated 2-(aminomethylene)benzo[b]thiophene-3(2Н)-ones with a terminal phenanthroline group
Beilstein J. Org. Chem. 2024, 20, 552–560, doi:10.3762/bjoc.20.47
- light and H+ or sequential addition of Fe2+ and AcO− ions. Keywords: fluorescence; molecular switches; N→O acyl rearrangement; naked eye effect; photochromism; Introduction Photochromism is defined as the reversible transformation of a molecular entity between different forms, having different
- inverse photochromism), such as merocyanine forms of spiropyrans and spirooxazines, azomethine imines, thioindigoid dyes and N→O acylotropic systems [12][13][14][15]. Recently, they have been actively used to create next-generation molecular switches, materials with new properties (in particular, color
- change depending on the intensity of sunlight), photochromic tags for biological research and optical sensors [15][16][17][18][19][20][21]. To develop new dual-mode molecular switches capable of efficient modulation of optical and fluorescent properties, both upon irradiation with visible light and upon
Switchable molecular tweezers: design and applications
Beilstein J. Org. Chem. 2024, 20, 504–539, doi:10.3762/bjoc.20.45

- Pablo Msellem Maksym Dekthiarenko Nihal Hadj Seyd Guillaume Vives Sorbonne Université, UMR CNRS 8232, Institut Parisien de Chimie Moléculaire, 4 place Jussieu, 75005, Paris, France 10.3762/bjoc.20.45 Abstract Switchable molecular tweezers are a unique class of molecular switches that, like their
- first part will be devoted to chemically responsive tweezers, including stimuli such as pH, metal coordination, and anion binding. Then, redox-active and photochemical tweezers will be presented. Keywords: coordination; molecular recognition; molecular switches; photoswitch; redox; supramolecular
- that binds to the OH groups instead of the pyrimidine nitrogen atoms. The group of Aprahamian has developed hydrazone-based molecular switches that can be controlled by photochemical or chemical stimuli such as pH [27]. They reported mesogenic tweezers-like compound 5 composed of a hydrazone switch
Synthesis, structure, and properties of switchable cross-conjugated 1,4-diaryl-1,3-butadiynes based on 1,8-bis(dimethylamino)naphthalene
Beilstein J. Org. Chem. 2023, 19, 674–686, doi:10.3762/bjoc.19.49
- , electron transfer, and quantum interference [17][18][19][20], but are also considered as promising molecular switches and transistors [21][22][23][24][25], NLO materials [26][27][28][29], and suitable starting compounds for syntheses involving multiple Diels–Alder additions [30]. All these facts motivated
Comparison of crystal structure and DFT calculations of triferrocenyl trithiophosphite’s conformance
Beilstein J. Org. Chem. 2022, 18, 1499–1504, doi:10.3762/bjoc.18.157

- properties. Such switchable systems with conjugated organic fragments containing an FeII/FeIII system were used in organic electronics as molecular switches, optoelectronic materials and in biochemistry as photonic or redox devices [6]. A promising approach is the coordination self-assembly of multiferrocene
Continuous flow synthesis of azobenzenes via Baeyer–Mills reaction
Beilstein J. Org. Chem. 2022, 18, 781–787, doi:10.3762/bjoc.18.78

- Azobenzene, as one of the most prominent molecular switches, is featured in many applications ranging from photopharmacology to information or energy storage. In order to easily and reproducibly synthesize non-symmetric substituted azobenzenes in an efficient way, especially on a large scale, the commonly
- . Keywords: azobenzenes; Baeyer–Mills reaction; continuous flow; molecular switches; solar fuel; Introduction Although the red-colored azobenzenes (AB) have been known for years as dyes, their applications nowadays span from energy and information storage [1][2][3][4][5], organocatalysis [6], photobiology
BINOL as a chiral element in mechanically interlocked molecules
Beilstein J. Org. Chem. 2022, 18, 508–523, doi:10.3762/bjoc.18.53

- applications as molecular switches, muscles, and motors [5][6][7][8][9][10][11], as novel materials [12], as medically active compounds [13][14], as catalysts [15][16][17][18][19], as chemosensors [20][21][22][23][24], and many more [25]. In view of their template-based synthesis and the importance of
Trichloroacetic acid fueled practical amine purifications
Beilstein J. Org. Chem. 2022, 18, 225–231, doi:10.3762/bjoc.18.26
- operations and the associated generation of waste, discovery of alternative purification techniques is mandatory. Avoiding waste accumulation, dissipative acids have recently found interesting applications in the field of out of equilibrium molecular switches and motors [5]. They enable the temporary
- cheap and simple acid enables a temporary protonation while time-controlled decarboxylation liberates volatile CO2 and chloroform as single waste (Scheme 2a). This strategy has been applied with success by the groups of Takata, Leigh, Kim and ours to time control different molecular switches ranging
- generated can be separated from the impurities. Closely related to out of equilibrium systems applied in molecular switches and motors under temporal control, from the amine salt, isolation of the pure free amine occurs through a simple decarboxylation of TCA, releasing volatile CO2 and CHCl3. As a result
Recent advances in the asymmetric phosphoric acid-catalyzed synthesis of axially chiral compounds
Beilstein J. Org. Chem. 2021, 17, 2729–2764, doi:10.3762/bjoc.17.185
- -catalyzed enantioselective transformations [9][14][22][23]. Axial chirality is also found in chiral stationary phases for enantioselective separation, dopants in liquid-crystalline materials, chiroptical molecular switches, microporous soluble polymers, and interlocked nanotubes (Figure 3) [24]. In addition
Construction of pillar[4]arene[1]quinone–1,10-dibromodecane pseudorotaxanes in solution and in the solid state
Beilstein J. Org. Chem. 2020, 16, 2954–2959, doi:10.3762/bjoc.16.245

- , including molecular switches, molecular logic gates, molecular machines, supramolecular polymers, etc. [6][7][8][9][10][11]. Pseudorotaxanes not only are used as the supramolecular precursors for the synthesis of rotaxanes and catenanes but also play an important role in the construction of supramolecular
Thermodynamic and electrochemical study of tailor-made crown ethers for redox-switchable (pseudo)rotaxanes
Beilstein J. Org. Chem. 2020, 16, 2576–2588, doi:10.3762/bjoc.16.209

- construction of more complex molecular structures [24][26][27]. With the introduction of stimuli-responsive units, crown ether/ammonium-based MIMs have evolved into molecular switches and motors [24][28]. Intriguing examples among them are a light-powered molecular pump [29], a chemical-fuel-driven molecular
- potential of tailor-made redox-active crown ethers for the development of new molecular switches. Yet, a careful design of tailor-made redox-active crown ethers is of great importance for tuning the crown ether binding and redox properties to achieve the desired molecular structure and switching mode, which
Synthetic approaches to bowl-shaped π-conjugated sumanene and its congeners
Beilstein J. Org. Chem. 2020, 16, 2212–2259, doi:10.3762/bjoc.16.186

- can not only be useful for enantioselective synthesis of π-bowls but also to produce novel building blocks for molecular switches, chemical machines, molecular motor, ferroelectric memories, molecular devices, and sensory materials etc. Interestingly, electronic switching, thermal transport and
Synthesis and highly efficient light-induced rearrangements of diphenylmethylene(2-benzo[b]thienyl)fulgides and fulgimides
Beilstein J. Org. Chem. 2020, 16, 1820–1829, doi:10.3762/bjoc.16.149
- ; X-ray diffraction; Introduction Photochromic compounds are of considerable interest as molecular switches, elements of optical memory and molecular logic devices [1][2][3][4]. Fulgides and fulgimides possessing excellent thermal and photochemical stability, structurally modulated fluorescent
- heteroaromatic fulgides revealed significant advantages of the latter for applications as molecular switches and optical information storage systems [5][10]. These include higher thermal stability, longer lifetimes of the photocolored forms and higher yields of the photocoloration reactions and better
Five-component, one-pot synthesis of an electroactive rotaxane comprising a bisferrocene macrocycle
Beilstein J. Org. Chem. 2020, 16, 1564–1571, doi:10.3762/bjoc.16.128
- to perform several functions as artificial molecular switches [1]. The template-directed synthesis of such sophisticated catenane and rotaxane molecular architectures allowed the expansion of chemical diversity and properties. Among these architectures, electroactive rotaxanes have been described to
Synthesis and characterization of bis(4-amino-2-bromo-6-methoxy)azobenzene derivatives
Beilstein J. Org. Chem. 2019, 15, 3000–3008, doi:10.3762/bjoc.15.296
- ; azonium; molecular switches; ortho substitution; photoisomerization; photoswitch; visible light; Introduction The application of photoswitches to control biological targets has been a driving force for the development of photoswitches that operate at wavelengths that are compatible with cells and tissues
Design, synthesis and investigation of water-soluble hemi-indigo photoswitches for bioapplications
Beilstein J. Org. Chem. 2019, 15, 2822–2829, doi:10.3762/bjoc.15.275

- that allowed to obtain an RNA-binding hemi-indigo derivative with photoswitchable fluorescent properties. Keywords: hemi-indigo; molecular switches; photochromism; photoswitching; visible light; water solubility; Introduction The application of organic photochromes in biological systems is fraught
A chiral self-sorting photoresponsive coordination cage based on overcrowded alkenes
Beilstein J. Org. Chem. 2019, 15, 2767–2773, doi:10.3762/bjoc.15.268

- between the three isomers. Two of the isomers were able to form host–guest complexes opening up new prospects toward stimuli-controlled substrate binding and release. Keywords: coordination cages; molecular motors; molecular switches; overcrowded alkene; palladium; Introduction Supramolecular
A combinatorial approach to improving the performance of azoarene photoswitches
Beilstein J. Org. Chem. 2019, 15, 2753–2764, doi:10.3762/bjoc.15.266

- results therefore define improved designs for high performance azo switches, which will allow for high precision optically addressable applications using such components. Keywords: arylazopyrazoles; azobenzenes; molecular switches; ortho-substitution; photoswitches; thermal half-life; Introduction
Experimental and computational electrochemistry of quinazolinespirohexadienone molecular switches – differential electrochromic vs photochromic behavior
Beilstein J. Org. Chem. 2019, 15, 2473–2485, doi:10.3762/bjoc.15.240
- focused on the preparation and investigation of electron-deficient analogs of the perimidinespirohexadienone (PSHD) family of photochromic molecular switches for potential application as "photochromic photooxidants" for gating sensitivity to photoinduced charge transfer. We previously reported the
- calculations of bond lengths, bond orders, and molecular orbitals allow the rationalization of this differential photochromic vs electrochromic behavior of the QSHDs. Keywords: cyclic voltammetry; density functional theory; heterocycles; molecular switches; photochromic photooxidants; spirocycles
- ; Introduction Photochromic molecular switches, in which light initiates reversible coloration of a short-wavelength isomer (SW) to a long-wavelength isomer (LW), which fades back to SW either thermally or photochemically, have become ubiquitous in a wide range of applications [1][2][3][4][5]. Typically, organic
Self-assembled coordination thioether silver(I) macrocyclic complexes for homogeneous catalysis
Beilstein J. Org. Chem. 2019, 15, 2465–2472, doi:10.3762/bjoc.15.239
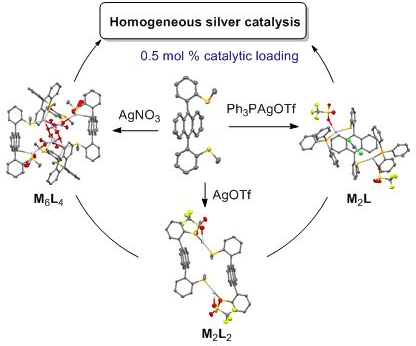
- ) to 25–29 kcal/mol (ortho-CH3). A few ortho-substituted DPA atropisomers have been described as potent molecular switches [48][49], building-blocks for self-assembled capsules [50][51] or optical materials [52][53][54]. However, due to their partially frozen structure, syn-isomers of DPA are ideal
Ultrafast processes triggered by one- and two-photon excitation of a photochromic and luminescent hydrazone
Beilstein J. Org. Chem. 2019, 15, 2438–2446, doi:10.3762/bjoc.15.236

- absorption cross-section of the molecule. Keywords: hydrazone; molecular switch; pump-probe spectroscopy; time-resolved fluorescence; Introduction Molecular switches are systems that are able to rapidly respond to an external stimulus, which can be of chemical or physical nature, through a variation of
Reversible switching of arylazopyrazole within a metal–organic cage
Beilstein J. Org. Chem. 2019, 15, 2398–2407, doi:10.3762/bjoc.15.232

- green light and resulted in the initial 1:2 cage/E-arylazopyrazole complex. This back-isomerization reaction also proceeded in the dark, with a rate significantly higher than in the absence of the cage. Keywords: arylazopyrazoles; coordination cages; inclusion complexes; molecular switches
- , the results reported here strengthen our understanding of the behavior of molecular switches in confined spaces. Experimental Synthesis of cage 2: Cage 2 was prepared based on a modified literature known procedure [18]. Briefly, a solution of [Pd(NO3)2(tmeda)] (200 mg, 0.577 mmol) in 25 mL of water
Targeted photoswitchable imaging of intracellular glutathione by a photochromic glycosheet sensor
Beilstein J. Org. Chem. 2019, 15, 2380–2389, doi:10.3762/bjoc.15.230

- therapy. Keywords: intracellular GSH; molecular switches; photochromic glycosheet; photoswitchable imaging; 2D MnO2 nanosheets; Introduction Cells are the basic structure and functional unit of biological organisms. Human diseases and aging are closely related to the states of cells. Thorough
Aggregation-induced emission effect on turn-off fluorescent switching of a photochromic diarylethene
Beilstein J. Org. Chem. 2019, 15, 2204–2212, doi:10.3762/bjoc.15.217

- Diarylethenes are well-known photochromic molecules and are widely applied to molecular switches and systems [1][2]. Recently, the photo-switching of fluorescence signals by using diarylethene switches has attracted much attention because of the potential in various optoelectronic applications [3]. For example
A heteroditopic macrocycle as organocatalytic nanoreactor for pyrroloacridinone synthesis in water
Beilstein J. Org. Chem. 2019, 15, 1505–1514, doi:10.3762/bjoc.15.152
- supramolecular assemblies like molecular rotors (pseudorotaxane, rotaxane, catenane), molecular switches, molecular shuttles, etc. [32][33][34][35][36][37][38][39][40][41][42][43]. Furthermore, macrocycles have been applied in the area of ion–ion pair recognition and heterometallic complex formation [44][45][46
Reversible end-to-end assembly of selectively functionalized gold nanorods by light-responsive arylazopyrazole–cyclodextrin interaction
Beilstein J. Org. Chem. 2019, 15, 1407–1415, doi:10.3762/bjoc.15.140

- –organic hybrid nanomaterials. Keywords: cyclodextrins; gold nanorods; host–guest chemistry; light-responsive materials; molecular switches; self-assembly; Introduction Metallic nanomaterials have received intense and interdisciplinary interest due to their unique optical [1], electronic [2][3] and
- sonication, whilst some dimers could not be fully dispersed. Possibly this observation can be explained from the limited photostationary states (PSS) of azobenzenes (E – Z: 80%, Z – E: 70%) [36]. Arylazopyrazoles (AAP) are a new class of molecular switches introduced by Fuchter et al. with excellent





































































































































































































































































































































































